“Despite the claims of various diet gurus, excess calorie consumption alone and not the amount of protein in an individual’s diet contributes to the accumulation of unwanted fat….” That’s the tendentious and pretty much inaccurate first line of the press release from JAMA on George Bray’s over-feeding study “Effect of Dietary Protein Content on Weight Gain, Energy Expenditure, and Body Composition During Overeating.” “Amount of protein?” What’s going here? It hasn’t really been about protein. Most of us “diet gurus” have claimed that carbohydrate, not protein, in the diet was the key macronutrient in regulating metabolism, consistent with the basic biochemistry of the glucose-insulin axis, or as Dr. Bray described Gary Taubes’s position in a review of Good Calories, Bad Calories:
“The problem is the carbohydrates in the diet, their effect on insulin secretion, and thus the hormonal regulation of homeostasis – the entire harmonic ensemble of the human body.”
Reduction in dietary carbohydrate puts increased demands on protein for gluconeogenesis and other processes but the controlling variable is the carbohydrate. The controversy in nutrition has been largely about fat vs carbohydrate. Should we be on a low-carbohydrate diet or a low-fat diet?
The quotation in the press release says accurately that “Earlier studies in human beings suggested that diets containing either high or low [levels of] protein are less ‘metabolically efficient’ than diets with normal protein levels.” Accurate, but written as if metabolic efficiency had always been recognized for its importance in weight loss, as if there had not been a dispute over whether the costs of processing protein were important in energy balance, indeed, written as if Bray and coworkers had not maintained that only calories count in weight gain or loss. The idea of metabolic advantage, that one diet could be more efficient — more weight gained/calorie — has been largely resisted by the nutritional establishment. Is this slouching toward Metabolic Advantage? (“Who knows not [the Duke] is dead? Who knows he is?”)
The debate is also about calories. Should you cut calories or just cut out carbs? Is it really “excess calorie consumption” and not the effect of excess carbohydrates ? “A calorie is a calorie” or not. Many of the gurus have gone beyond “claiming” to demonstrating that when carbohydrates are low, weight loss is greater than when carbohydrate is high and that the weight loss on a low-carbohydrate diet is primarily in fat stores rather than lean mass. In head-to-head comparisons, for however long they are compared, low-carbohydrate diets generally out-perform low-fat diets on other parameters as well, glycemic control, the features of atherogenic dyslipidemia. This has been the major challenge to traditional nutrition and the general approach has been to simply ignore this data and dismiss the researchers with innuendo as above.
In some sense, Bray, et al. answered a question that we weren’t asking, but protein is important if more complicated than carbohydrate and fat. So what did the study find? Bray and coworkers compared three diets of 5 %, 15 % and 25 % protein at an excess of calories, that was nominally the same in each group. The study was a random controlled study and was carried out in a metabolic ward so the results are more accurate than the usual diet study that relies on dietary records. There is something odd about this study, though, in that if you want to say that only calories are the independent variable, you can’t keep calories constant. What was actually done was to determine the energy requirements for weight maintenance over a run-in period of 2-3 weeks on a maintenance and then an additional 40 % of calories was added. So although the calories are constant relative to initial energy expenditure, they are not absolutely the same and this is a study of the effect of varying calories while keeping calories constant. The figure below, re-drawn from Figure 6 of the paper comparing intake of absolute energy to protein intake makes you stop and think.

When you have a small number of subjects, a single outlier can bias the results. If you remove the single highest point (circled in red), the correlation is likely to get much weaker and the normals and low begin to separate. In other words, the individual variation (the relative efficiency) is sufficient to make it hard to see the effect of variable energy or, perhaps, as the authors themselves set it up, it is energy normalized for baseline that is the key variable. Then the authors are right (at least by inspection) that the protein intake does not effect the change in body fat but you have only a single value for the energy. In this case, you cannot say “calories alone account for the increase in fat” (Conclusion in Abstract) because you have only a single point. If you keep constant the variable (carbohydrate) that is most likely to bring out differences, you shouldn’t be surprised in there are no big differences.
Even taking the conclusions at face value, the authors found, as other diet comparison studies have, that weight loss or, in this over-feeding study, weight gain, was not dependent on calories alone: “a calorie is a calorie” not. It is likely that this was what the study was originally trying to disprove and the results must have been a disappointment. The way out was that, in this particular case, the differential weight loss showed up in difference in lean mass, rather than in fat mass as has been found in other studies showing variable efficiency. Since 5 % is very low protein it is probably not surprising that the diet could not provide enough protein for an increase in lean mass this group.
So what are the other diet studies that have found variable efficiency. The reduction in weight found in studies comparing low-carbohydrate diets and low-fat diets not only shows a difference favoring carbohydrate restriction but the improved weight loss is preferentially fat over lean mass. For example, Volek, et al. compared a low fat with a VLCK and the results are as shown below. In their study, subjects were randomized to one of two hypocaloric diets, a very low-carbohydrate ketogenic (VLCK) diet (carbohydrate <10% of energy) or a low fat (LF) diet and after 8 weeks switched to the other diet. Reported energy was slightly higher during the VLCK but the VLCK group lost more weight and as shown below predominantly in fat, total fat loss, and trunk fat loss for men (despite significantly greater energy intake). The majority of women also responded more favorably to the VLCK diet, especially in terms of trunk fat loss the ratio of trunk fat/total fat was also significantly reduced during the VLCK diet in men and women. These studies depend on diet recall so are less accurate than the JAMA study but because of the better experimental design, the changes are bigger and with appropriate correction make a less ambiguous case than the JAMA study. The more accurate measurements in the metabolic chamber suggest that individual variation is real and not just due to random error.

So what do we know from Bray, et al.? As described above, there is some ambiguity in what constant energy means. Still, nobody questions that under many conditions, a “calorie is a calorie,” but they actually found that weight gain was different so when metabolic advantage is “claimed” it cannot be dismissed out of hand. This is different than widely cited studies in the literature that claim macronutrients do not effect weight loss, since if weight gain depends on macronutrient, it is reasonable that weight loss does too. Similarly, if tissue distribution affects lean mass in this case, then studies where the tissue distribution shows preferential loss of fat can’t be dismissed — again, it is certainly not surprising that a low protein diet will lead to less storage of protein; generally, while it is just as bad a generalization as “a calorie is a calorie,” there is some truth in “you are what you eat.” Also, in the JAMA study, protein was exchanged for fat so a reduction in fat did not have an effect on fat which may or may not be a surprise to many people. Tom Naughton raised a few other questions about Bray, et al. but in the end, the paper reminds me of the joke about the Polish Mafia: they make you an offer you can’t understand.
How to do it.
But I told George how to do it. A couple of years ago, he and I had a brief correspondence. I made the following proposal. I suggested we could apply for a joint grant and publication to get the answer. The following is from my email to him in 2008 (I have added some highlights):
“A modest proposal
Proponents of carbohydrate-restricted diets (CRD) and critics of such diets cooperate to design a long-term comparison of CRD and low-fat diets. The groups agree on methods of procedure, make-up of the diets, how compliance will be effected, and what parameters will be measured.
We write the paper first, leaving room for the data, that is, we agree in advance on what the possible outcomes are and what conclusions could be drawn from them. The final MS can only be edited for language usage. There are no disclaimers, no Monday-morning-quarterbacking, no excuses.
The paper could be submitted while the grant application is being written and would have to be accepted because any objections could be incorporated in the plan. The grant itself would surely be funded since it incorporates everybody’s specific aims.”
George hasn’t answered and he obviously has a different approach to the problem but my offer still stands.
In the end, that is what it will take to solve the problem. Unless we agree on what the question is, how it can be tested and work together to do the experiment, the lipophobes will ignore the low-carbohydrate studies and we will criticize their studies. The real losers, of course, will be the people suffering from obesity and diabetes. The question everybody always asks me, is why can’t there be a meeting of the minds? In the current case, why was the JAMA study done?
Why was this study done?
“Dr Bray discussed the results with news@JAMA via e-mail.
news@JAMA: What are the practical implications of these findings for patients trying to lose weight or for the physicians trying to counsel them?
Dr Bray: The first practical implication is an old one: calories count. We showed very clearly that the increase in body fat was due to the increased intake of calories and that the amount of protein in the diet did not change it.
To avoid that slow weight gain that many adults experience in their middle years, people need to watch their weight and increase activity, decrease food intake, or both; changing the diet alone will not do it.”
This sounds like the the same recommendations we’ve had for years. Writing this, I suddenly realized that, as they say in German: that’s where the dog is buried. It is about recommendations. This research is following the recommendations. It used to be (should be? assume it must be?) that recommendations follow from the research. Now, it’s the other way around. Committees make recommendations and then research (sometimes by members of the committee) tries to support the recommendations. Something about this bothers me.

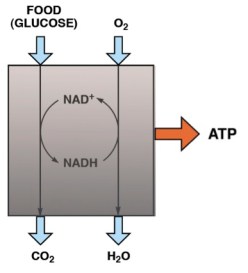
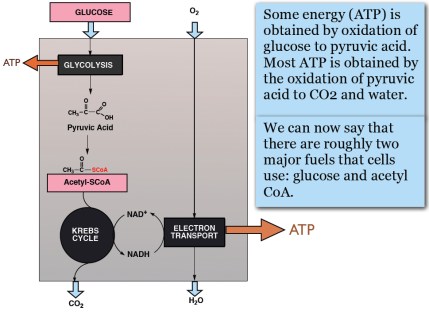
 There is thus a steady-state that continuously readjusts levels of fat and fatty acid. The process will drift in the direction of oxidation when stored fat provides energy to other cells and will tend in the opposite direction — toward synthesis — when fat is stored. The important point is that the steady-state, like an equilibrium state, does not mean that everything has stopped. It means that the forward rate of breakdown is equal to the resynthesis rate. Every time there is a cycle, TAG → FA → TAG, however, energy is wasted — synthesis of TAG requires ATP, lipolysis is spontaneous and no ATP is re-syntesized. Why would such a thing evolve? The common name of the process is substrate cycle but because each cycle wastes ATP and accomplishes nothing — you get back the substrate that you started with — it has been referred to as a “futile cycle.” Why would the adipocyte waste energy in this way?
There is thus a steady-state that continuously readjusts levels of fat and fatty acid. The process will drift in the direction of oxidation when stored fat provides energy to other cells and will tend in the opposite direction — toward synthesis — when fat is stored. The important point is that the steady-state, like an equilibrium state, does not mean that everything has stopped. It means that the forward rate of breakdown is equal to the resynthesis rate. Every time there is a cycle, TAG → FA → TAG, however, energy is wasted — synthesis of TAG requires ATP, lipolysis is spontaneous and no ATP is re-syntesized. Why would such a thing evolve? The common name of the process is substrate cycle but because each cycle wastes ATP and accomplishes nothing — you get back the substrate that you started with — it has been referred to as a “futile cycle.” Why would the adipocyte waste energy in this way? Diesel engines
Diesel engines .
.




 The car analogy of metabolic inhibitors. Figure from my lectures. Energy is generated in the TCA cycle and electron transport chain (ETC). The clutch plays the role of the membrane proton gradient, transmitting energy to the wheels which produce forward motion (phosphorylation of ADP). Uncouplers allow oxidation to continue — the TCA cycle is “racing” but to no effect. Other inhibitors (called oxidative phosphorylation inhibitors) include oligomycin which blocks the ATP synthase, analogous to a block under the wheels: no phosphorylation, no utilization of the gradient; no utilization, no gradient formation; no gradient, no oxidation. The engine “stalls.”
The car analogy of metabolic inhibitors. Figure from my lectures. Energy is generated in the TCA cycle and electron transport chain (ETC). The clutch plays the role of the membrane proton gradient, transmitting energy to the wheels which produce forward motion (phosphorylation of ADP). Uncouplers allow oxidation to continue — the TCA cycle is “racing” but to no effect. Other inhibitors (called oxidative phosphorylation inhibitors) include oligomycin which blocks the ATP synthase, analogous to a block under the wheels: no phosphorylation, no utilization of the gradient; no utilization, no gradient formation; no gradient, no oxidation. The engine “stalls.”