“I may have killed a dozen men but I never stole a horse.”
— last words of outlaw in the American West before being hanged.
The principle known as Occam’s Razor is usually understood as a statement that a simple explanation is preferable to one that is more complicated. The principle has many variations. It might be interpreted as saying that you have to have a sense of priorities. Occam’s Razor is not exactly a scientific idea so much as a principle of aesthetics expressing the value of elegance in scientific explanations. Named for
William of Ockham (c. 1285–1349) — it is also referred to as Ockham’s Razor — the idea can be described mathematically by saying that if the outcome,
Y, of an experiment can be expressed with a rough sort of equation:
Y =
A +
B +
C +… and if
A explains
Y, then you don’t want to drag in
B,
C, etc unless you absolutely have to. (A more compelling description might be to consider the principle in terms of a power series and if you are inclined to mathematics,
Wikipedia has excellent description and animation).
 Where we’re going. The bottom line on this post is that for obesity, diabetes and general health, the predominant effect of diet, the major contribution to the outcome — A in the equation above — is provided by substituting fat (any fat) for carbohydrate (any carbohydrate). That’s what the science says. That will give you the best effect. The B contribution (type of fat, type of carbohydrate) is strictly secondary. The practical consequence: if for some reason, you want to reduce fructose in the diet, the best advice is to reduce carbohydrate across the board. You can then add the additional advice “preferably sugar and high fructose corn syrup” but even if B doesn’t kick in, you will surely get a benefit. Most of all, if you take out Pepsi® and put in Pepperidge Farm® Whole Wheat Bread, you may not accomplish much.
Where we’re going. The bottom line on this post is that for obesity, diabetes and general health, the predominant effect of diet, the major contribution to the outcome — A in the equation above — is provided by substituting fat (any fat) for carbohydrate (any carbohydrate). That’s what the science says. That will give you the best effect. The B contribution (type of fat, type of carbohydrate) is strictly secondary. The practical consequence: if for some reason, you want to reduce fructose in the diet, the best advice is to reduce carbohydrate across the board. You can then add the additional advice “preferably sugar and high fructose corn syrup” but even if B doesn’t kick in, you will surely get a benefit. Most of all, if you take out Pepsi® and put in Pepperidge Farm® Whole Wheat Bread, you may not accomplish much.
In practical terms, confronted with a phenomenon that has many controlling variables, make sure you can’t do with one before you bring in the others. In nutrition, when people say that the phenomenon is very complicated, they frequently mean that they don’t want to look at a simple explanation. On its practical side, if a patients in a dietary experiment responds to the level of carbohydrate, you have to assume that carbohydrate across the board is the controlling variable. If, however, you think that it is specifically the fructose in the diet that caused the effect, or if you think that it was an additional effect of fructose beyond its role as carbohydrate, then that is something that you have to show separately. Until you do, the fructose effect is sliced off by Occam’s Razor. In terms of policy, you don’t want to go after fructose unless you are sure that it is not primarily the role of fructose acting as a carbohydrate.
So, there is a logical question surrounding recommendations against sugar and especially against fructose. What we know well in nutrition is that if you replace carbohydrate with fat, as in Krauss’s experiment described in the previous post, things improve and this is why we suggest low-carbohydrate diets as the “default diet,” the one to try first for diabetes and metabolic syndrome and probably for cardiovascular risk. I have, however, received at least two emails from well-known nutritionists saying that “the type of carbohydrate is more important than how much carbohydrate” and, of course, Rob Lustig is everywhere telling us how toxic sugar is but never suggesting that a low carbohydrate diet is any kind of ideal. On the face of it, the idea doesn’t make much sense. Fructose is a carbohydrate so the amount and type are not easily separable.
There are all kinds of strange things in nutrition. People actually say that the type of diet you are on is less important than whether you stay on the diet. While true, it is like saying that if you are baseball player, whether you get a hit depends less on who’s pitching than whether you remember to show up for the game. But anyway, I decided to ask the question about relative importance of type and amount of carbohydrate on facebook and on a couple of blogs where things like Hizzona’ Michael Bloomberg’s Big Bottle Ban or related questions was being discussed. Here’s how I put it.
For general health, should you change the type of carbohydrate or replace the carbohydrate with fat (any natural fat, no trans-fat)? It’s a thought experiment (not real world situation with subtleties). You only get three choices: For general health (no change in calories):
1. Change type of carbohydrate
2. Replace carbohydrate with fat
3. It doesn’t matter
Strangely enough, I did not get very many answers. I think that people didn’t like the question and even when they voted, they wanted to put in disclaimers:
ANS: 2. Replace carbohydrate with fat But I want to add; not replacing ALL the carbs. Only the worst ones. You know; Sugar, grains (bread and pasta) potatoes and rice.
RDF: You can do that in a real case but the question is about first-order strategies. You only get 3 choices.
ANS: okej 2. Replace carbohydrate with fat.
And James Krieger jumped in:
“Feinman, your ‘thought experiment’ is essentially a false trichotomy…same thing as a false dichotomy except you’ve arbitrarily limited it to 3 choices rather than 2, when in fact there are many more. This is why you aren’t getting answers…because you’re committing a common logical fallacy.”
I explained that
“It’s called Occam’s Razor…. I’m simply asking: if you could theoretically do only one thing, 1. or 2., which would be better? There are many other choices but in a thought experiment you imagine these to be held constant or to be the higher order terms in a power series.”
But, of course, Krieger was right. The question is not really answerable. Not because it is false so much as because it is confused. Fructose is a carbohydrate and whatever its unique contribution, it is hard to say it is more important than the contribution of the fructose as a carbohydrate. It is a screwy idea but, again, that’s how it was phrased to me in emails and probably in print someplace. Researchers in this field say: “it is not carbohydrate per se (or glycemic index/load) that is involved in adverse metabolic effects of dietary carbohydrates, but rather the type of carbohydrate,…” The kind of evidence that is used to support such an idea, the kind of result that is used to support fructophobia is in the paper by Stanhope, et al.
Stanhope, et al. measured the effects of chronic consumption of either glucose- or fructose-sweetened beverages providing 25% of energy requirements for 10 weeks in overweight and obese subjects. The figure below shows the superimposed outcomes in the response of triglycerides in the course of a day (red lines = fructose, blue = glucose). It is obvious that there is a difference — people consuming fructose had higher triglyceride responses (although fasting levels were not different). Looking at the figure, though, there is big variation in the data and it is not clear that everybody showed big differences between the glucose and fructose curves: the error bars represent standard error of the mean (SEM) which, while it shows you that there may be a statistically significant difference between the trials, doesn’t display very well the spread of the individual values, that is, whether a few individuals biased the grouped data. To convert to standard deviation, which gives you a better feel for the variation, you multiply, in this case, by about 4. In other words, there must have been big overlap between the fructose people and the glucose people.
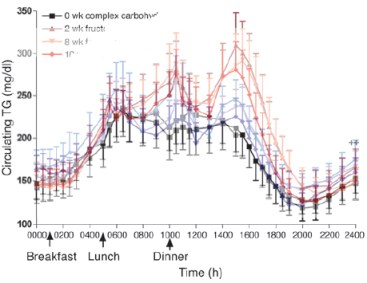
So there is an effect of type of carbohydrate. But what to compare it to? The study of Krauss in the previous post showed much bigger changes when you substituted fat for carbohydrate and, in fact, those were fasting triglycerides which, in the fructose experiment, didn’t change at all but this is a different kind of experiment. So for comparison, we can look at a study from Jeff Volek’s lab where carbohydrate was replaced with fat in the carbohydrate restricted diet (CRD) in comparison to a low-fat diet (LFD). I described this study previously because it showed how carbohydrate, rather than dietary saturated fat, was actually controlling saturated fat in the blood. Here is what the responses to meals as seen in plasma triglycerides:
 Maybe it’s the Fructose.
Maybe it’s the Fructose.
The fructose experiments can be shaved with Occam’s razor — insofar as we can tell, reducing carbohydrate across the board is more effective than changing type of carbohydrate. But how do we know that the effect of reducing carbohydrates wasn’t due to removing fructose — fructose is a carbohydrate so carbohydrate restriction may be due to the de facto removal of the fructose? Well, we don’t. It’s unlikely but possible. Where does this leave us? Wikipedia cites Bertrand Russell’s variation of Occam’s Razor: “Whenever possible, substitute constructions out of known entities for inferences to unknown entities.” This is a pompous way of saying: “don’t make things up.”
Another way of looking at Stanhope’s experiment is to recognize that it does not show, as the title says, “Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids… in overweight/obese humans.” What the paper really is about is “Consuming fructose-sweetened, not glucose-sweetened, beverages as part a high carbohydrate diet (55 % of energy) increases….” In other words, you don’t know whether you would get any benefit in changing from fructose to glucose if the total carbohydrate were lower. In terms of our Occam’s Razor equation, you can’t say that you have proved that your results are due to A (the major controlling variable (carbohydrate)) when all you have studied is A with the specific change in the B term (secondary effect of the type of carbohydrate). Stanhope’s experiment shows: if you are on a high carbohydrate diet, replacing glucose with fructose will make things worse but that’s different than saying that fructose is toxic. From a practical point of view, if you are on a high carbohydrate diet and it is not giving you the health benefit you want, replacing sugar with starch may give you disappointing results compared to simply cutting down on carbohydrates.
How to Reduce Fructose Consumption.
If you want to encourage fructose reduction, encourage carbohydrate restriction (this is where we have the most information) with the additional proviso of recommending fructose reduction as the first carbohydrate to remove (may also help but we have less data).
Flawed Studies.
In combination with the previous post, a summary of things to look for in a study to make sure that the authors are not misleading you and/or themselves:
1. Understatement is good. “Healthy” is a value judgement. “Fructose-sweetened” is not the same thing as “fructose-sweetened in a high carbohydrate diet.”
2. Where are the pictures? The author has an obligation to make things clear. A graphic representation is usually an indication of a desire to explain.
3. Has Occam’s Razor been applied? Are secondary effects taken as primary?
Crabtree’s Bludgeon
Finally, we should not forget Crabtree’s Bludgeon which is described by Wikipedia as “a foil to Occam’s Razor” and “attributed to the fictitious poet, Joseph Crabtree, after whom the Crabtree Foundation is named.” It may be expressed as:
‘No set of mutually inconsistent observations can exist for which some human intellect cannot conceive a coherent explanation, however complicated.’